Interactive approach establishes a well-deserved academic connect between you and Master Teachers. Sessions get recorded for you to access for quick revision later, just by a quick login to your account. Your academic progress report is shared during the Parents Teachers Meeting. Assignments, Regular Homeworks, Subjective & Objective Tests promote your regular practice of the topics. Revision notes and formula sheets are shared with you, for grasping the toughest concepts. WAVE platform encourages your Online engagement with the Master Teachers. We provide you year-long structured coaching classes for CBSE and ICSE Board & JEE and NEET entrance exam preparation at affordable tuition fees, with an exclusive session for clearing doubts, ensuring that neither you nor the topics remain unattended. We have grown leaps and bounds to be the best Online Tuition Website in India with immensely talented Vedantu Master Teachers, from the most reputed institutions. 
Vedantu LIVE Online Master Classes is an incredibly personalized tutoring platform for you, while you are staying at your home. 
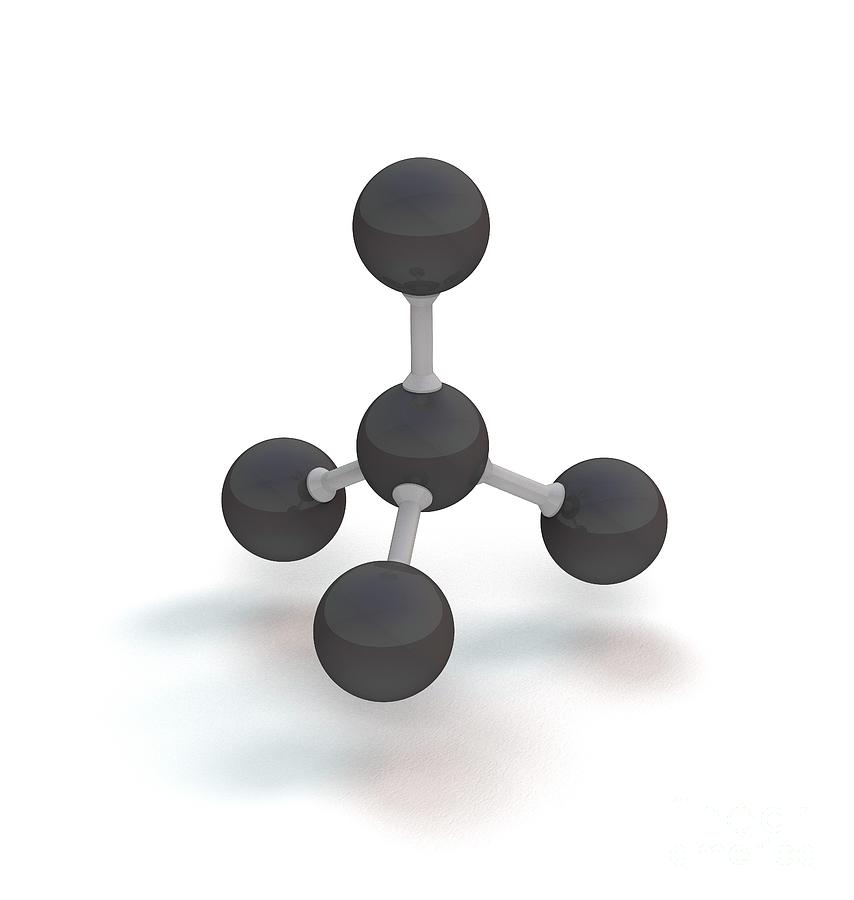
The atoms in a molecule of water adopt what kind of geometry? Which of the following molecules has tetrahedral geometry?Ģ. The word "tetra" means "four," and the word "hedral" represents a solid face.Ī central atom is located in the centre of a tetrahedral molecular geometry, with four substituents located at the corners of the tetrahedron.ġ. The word "tetrahedral" gives us a good idea of what this term means. 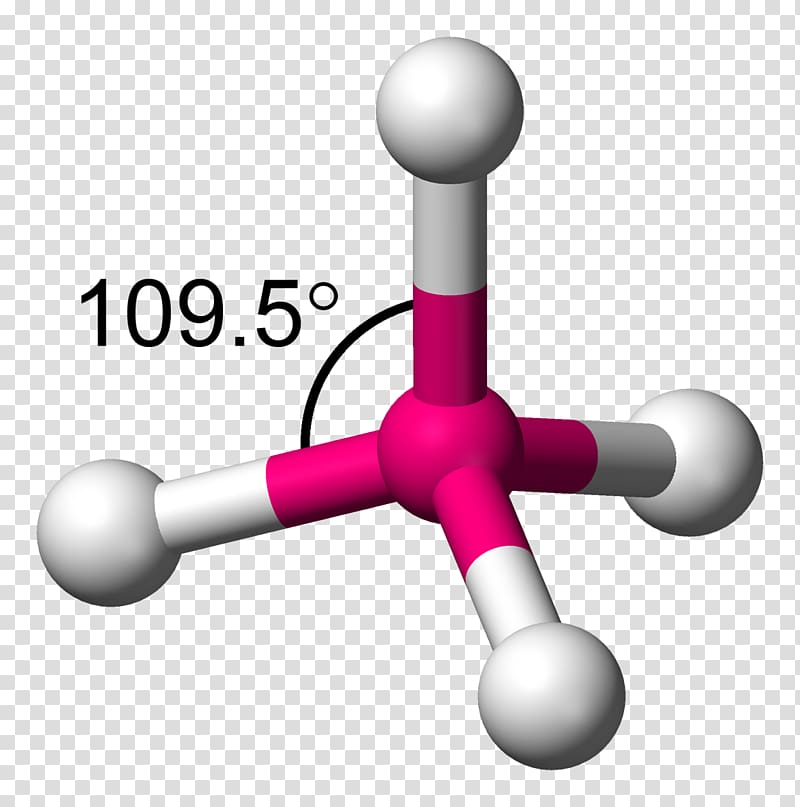
The tetrahedral shape is formed when four atoms in their elemental form covalently bond together. Of course, there are many more examples in each category in fact, there are far fewer cases of purely non-polar substances in each of these categories than polar substances. Analysis of the distribution of the electron density suggests that the larger value of C-Ge-C in Me(2)GeCl(2) compared to the equivalent but smaller angle in Me(2)CCl(2) is indirectly the result of the higher ionic character of the bonds in the former molecule.A central atom is located in the centre of a tetrahedral molecular geometry, with four substituents located at the corners of the tetrahedron. Calculations show that the conformation of the organic substituents has a sizable effect on the local geometry of the Ge-atom. An experimental value of 121.2 +/- 0.2 degrees is found for the C-Ge-C angle in the crystal structure of dichlorodi(2-phenethyl)germane, and it provides the first crystallographic evidence for the deviation from tetrahedral geometry. The results show that, in the crystal, intermolecular interactions do have a large influence on the geometry of the molecule. The molecular structure of Me(2)GeCl(2), and the value of the C-Ge-C angle in particular, was studied by ab initio quantum calculations to examine the deviation of this molecule from ideal geometry in the gas phase and in the crystalline state.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
